Ozone lamps
_UV lamps are energy source for both making and destroying ozone.
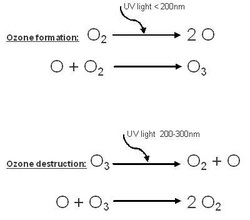
Ozone formation: When a photon of light with a wavelength shorter than 200 nanometers is absorbed by an Oxygen molecule (O2), the energy breaks the molecule into two Oxygen atoms (O). One of these atoms can join with an Oxygen molecule to form an Ozone molecule (O3).
Ozone destruction: When a photon of light with a wavelength between 200 to 300 nanometers is absorbed by an Ozone molecule (O3), the energy breaks the molecule into an Oxygen molecule (O2) and an Oxygen atom (O). The Oxygen atom can break an Ozone molecule and form two Oxygen molecules.
Ozone generation using ultraviolet light has been used for many years for many industrial applications. Ozone is a powerful oxidizing agent. It can oxidize many organic compounds and is used commercially as a bleach for waxes, oils, textiles, and as a deodorizing agent. Because it is a powerful germicidal agent, it is also used to sterilize air and drinking water. The amount of the ozone produced by a UV lamp depends on the application design, but it is also greatly impacted by the ambient temperature, humidity, and the air flow speed near the lamp.
Ozone Applications
Ozone formation: When a photon of light with a wavelength shorter than 200 nanometers is absorbed by an Oxygen molecule (O2), the energy breaks the molecule into two Oxygen atoms (O). One of these atoms can join with an Oxygen molecule to form an Ozone molecule (O3).
Ozone destruction: When a photon of light with a wavelength between 200 to 300 nanometers is absorbed by an Ozone molecule (O3), the energy breaks the molecule into an Oxygen molecule (O2) and an Oxygen atom (O). The Oxygen atom can break an Ozone molecule and form two Oxygen molecules.
Ozone generation using ultraviolet light has been used for many years for many industrial applications. Ozone is a powerful oxidizing agent. It can oxidize many organic compounds and is used commercially as a bleach for waxes, oils, textiles, and as a deodorizing agent. Because it is a powerful germicidal agent, it is also used to sterilize air and drinking water. The amount of the ozone produced by a UV lamp depends on the application design, but it is also greatly impacted by the ambient temperature, humidity, and the air flow speed near the lamp.
Ozone Applications
- Disinfection of laundries in hospitals, food factories, senior care facilities etc;
- Disinfection of water
- Deodorize air and objects, treat smoke damage. This process is extensively used in fabric restoration after fire damage or excessive indoor smoking.
- Inactivate bacteria on food or on contact surfaces
- Surface treatment of eggs and fruit
- Sanitize swimming pools and spas
- Scrub yeast and mold spores from the air in food processing plants
- Wash fresh fruits and vegetables to inactivate yeast, mold and bacteria
- Chemically attack contaminants in water (iron, arsenic, hydrogen sulfide, nitrites, and complex organics lumped together as "color");
- Provide an aid to flocculation (agglomeration of molecules, which aids in filtration, where the iron and arsenic are removed);
- Manufacture chemical compounds via chemical synthesis
- Clean and bleach fabrics
- Assist in processing plastics to allow adhesion of inks;
- Age rubber samples to determine the useful life of a batch of rubber;
- Eradicate water borne parasites such as Giardia and Cryptosporidium in surface water and waste-water treatment plants
- Aquaculture, fish farming
- Pathogen reduction in cooling towers
